FDA recalls batch on injection after suspected death to reaction in Maharashtra:
Thu 24 Nov 2022, 09:15:39
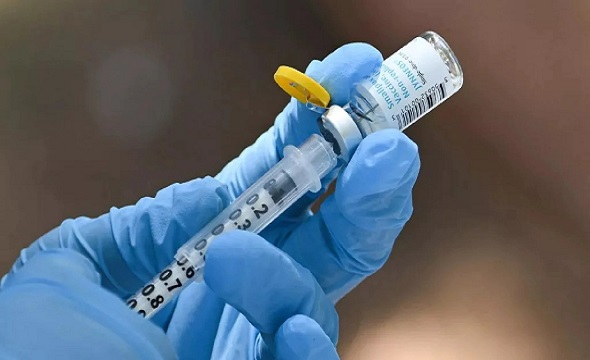
Mumbai: The Maharashtra Food & Drug Administration (FDA) has asked all drug controller authorities in the country to stop the use of a particular batch of the Orofer FCM injection manufactured by Emcure Pharmaceuticals Ltd from across the country. This comes after a 55-year-old male patient died last month in Saifee Hospital at Charni Road due to a suspected adverse reaction to the drug.
“We received a complaint from the wife of the deceased on November 4 stating that her husband had died on October 12 after a suspected adverse drug reaction. The patient was given Orofer FCM (Ferric Carboxymaltose Injection 500 mg /10 ml) which is used to treat iron-deficiency anaemia in adults who can't be treated with iron supplements taken orally,” said an official.
A team of drug inspectors visited the Saifee Hospital on November 10 and collected the remaining seven vials which
were sent for testing. FDA joint commissioner (drugs) SB Patil said Emcure was asked to recall the particular batch of the drug. “According to the company, there could be spurious drugs available in the market in the name of INJ OROFER and because of such spurious drugs the suspected adverse reaction might have taken place,” he said.
were sent for testing. FDA joint commissioner (drugs) SB Patil said Emcure was asked to recall the particular batch of the drug. “According to the company, there could be spurious drugs available in the market in the name of INJ OROFER and because of such spurious drugs the suspected adverse reaction might have taken place,” he said.
According to Mr Patil, the company has told the FDA that spurious versions of one of its other drugs is already being sold in the market. “The Mumbai FDA team is carrying out a probe in the matter,” Mr Patil said, adding that samples from the batch of the drug have been collected from the distributors and sent for analysis.
“We need at least 40 samples to reach a conclusion. The Pune FDA has also collected a sample of the same batch and sent it for testing. We expect a report within two weeks,” he concluded.
No Comments For This Post, Be first to write a Comment.
Most viewed from General
Most viewed from World
AIMIM News
Latest Urdu News
Most Viewed
May 26, 2020
Which cricket teams will reach the IPL 2026 finals?
Latest Videos View All
Like Us
Home
About Us
Advertise With Us
All Polls
Epaper Archives
Privacy Policy
Contact Us
Download Etemaad App
© 2026 Etemaad Daily News, All Rights Reserved.

































.jpg)
.jpg)
.jpg)


